Our Chemical Protein Synthesis Platform
Platform Generates Conjugatable Cytokines With Optimized Properties
Our platform has the unique capability to overcome limitations of bacterial and cellular protein expression systems. The technology allows us to construct novel proteins, such as cytokines, with optimized pharmacological properties – rationally designed for improved safety, enhanced efficacy, and with broader utility.
Bright Peak’s chemical synthesis method provides unlimited flexibility to both fine-tune and enhance biological functions through affinity modification and orthogonal, site-specific conjugation at any desired site in the protein. This is enabled by our novel α-ketoacid-hydroxylamine (KAHA) ligation technology which allows us to chemically ligate smaller, precisely customized peptide segments together to create therapeutic proteins with deliberately refined pharmacological properties.
Bright Peak cytokine payloads:
Utilizing master cytokines – powerful master regulators
of immune responses
Optimized affinity & selectivity
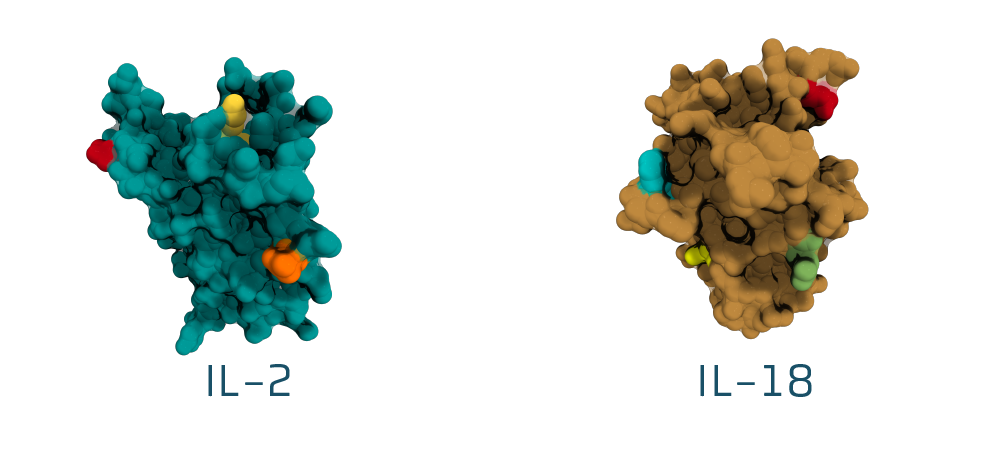
Enhanced therapeutic utility through site-specific chemical conjugation
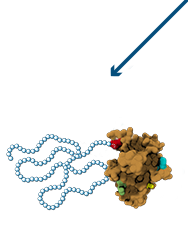
Best-in-Class Single Agents
- Rationally Designed
- Enhanced PK
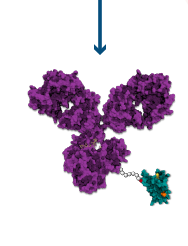
Bright Peak Immunoconjugates –
Pioneering a New Class of Biologics
- Proprietary and simple ADC-like technology
- Conjugatable to existing mAbs without structural modifications – “Plug and Play” flexibility
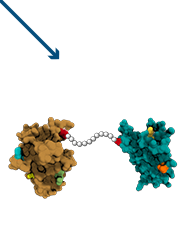
Duokines –
The New Cytokine Frontier
- Cytokine-cytokine conjugation creates novel biology
- Potential for synergy
Bright Peak Immunoconjugates – The Next Generation of Precision Immunotherapy
Chemical synthesis allows Bright Peak’s cytokines to be engineered as ‘payloads’ that can be easily conjugated through a proprietary linker technology to a diverse array of biological molecules, such as monoclonal antibodies (e.g., anti-PD-1), creating a novel class of biologics we call “Bright Peak Immunoconjugates”.
One of the many advantages of our technology platform is the ‘off-the-shelf’ ability to convert existing therapeutic antibodies into dual-targeting, multi-functional immunoconjugates by a simple and rapid chemical process. As a result, Bright Peak’s proprietary approach allows the use of antibodies ‘as-is’, avoiding the complex process of generating recombinant antibody-cytokine fusion proteins which involves lengthy protein expression, optimization and cell-line development.
Our multi-functional immunoconjugates for cancer reflect the latest insights in immune checkpoint and cytokine biology. We utilize monoclonal antibodies, such as anti-PD-1, to target our enhanced cytokines to specific immune cell populations in the tumor, and to simultaneously deliver the combined effects of checkpoint blockade and cytokine agonism to these cells (“cis-signaling”). In doing so, we believe that this approach can more precisely and effectively engage the immune response within tumors, with the opportunity for improved biologic activity, safety and efficacy.
Upgrading Immune Checkpoint Therapy to “PD-1 2.0”
Our unique immunoconjugate platform is designed to upgrade immune checkpoint therapy to the next level, “PD-1 2.0”, and potentially establish a new backbone in immuno-oncology.
Our novel, multifunctional PD-1-based immunoconjugates target and block the dominant inhibitory immune checkpoint pathway (PD-1/PD-L1) and simultaneously deliver our functionally-enhanced master regulatory cytokines (IL-18 or IL-2) in cis to select immune cell populations in the tumor microenvironment. Our next-generation “PD-1 2.0” agents are designed to selectively target and activate antigen-experienced, PD-1+ CD8+ cytotoxic T cells while avoiding the broad activation of other immune cells. Cis-signaling achieved uniquely by PD-1-based immunoconjugates can expand stem-like effector T cells that demonstrate superior anti-tumor efficacy in preclinical models and have been associated with improved outcomes to checkpoint inhibition in patients.
Key Attributes to our Platform
- Pioneering “Bright Peak Immunoconjugates”, a new class of multi-functional biologics
- Bright Peak Immunoconjugates are comprised of optimized cytokines chemically conjugated to existing antibodies – proprietary “off-the-shelf” technology – applicable to the development of novel therapeutics for both cancer and autoimmune disease
- Optimized cytokine payloads are generated using our proprietary chemical synthesis and KAHA ligation technology platform
- Site-specific modification using chemistry provides full control of cytokine characteristics, including introduction of conjugation handles at any position – technology uniquely positioned to leapfrog 1st generation recombinant fusion protein technologies
- Novel Intellectual Property space

 '
'